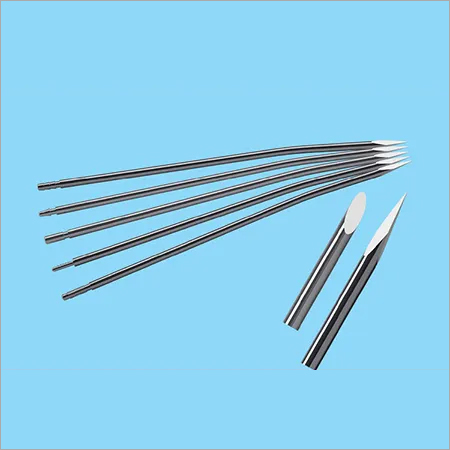
We are the leading Manufacturer, Supplier & Exporters of Wound Drainage Instruments and Drainage Trocar.
Best Seller
Best Seller
About Us
Bio Medical Devices is the leading manufacturer and exporter of SS Trocar, Wound Drainage Instruments, SS made Disposable Close Wound Drainage Trocars in different sizes for medical applications in close wound suction unit. We are supported by a team of talented personnel having rich experience and have a well fitted Research and Development facility which enables us to offer quality and innovative products to our patrons. We are an ISO 9001:2000 accredited company so our clients can be assured about the quality of products.
Combining advanced technology with quality, we take pride in introducing ourselves as Exporter, Manufacturer and Distributor of SS Trocar in different sizes FG-6, FG-8, FG-10, FG-12, FG-14, FG-16, FG-18, FG-20, FG-22, FG-24, FG-26 and FG-28. We have been successfully in serving and supplying these Trocars to various ISO certified and CE marked companies in India and through direct export to various countries across the globe.
Trocar: It is a sharp and pointed shaft with three sided point. It can be used with a cannula, a hollow tube which is designed to be inserted in vein, bone marrow, artery or body cavity. Trocar is derived from French trois (three) + carre (side). It is a three side hollow cylinder with a sharply pointed end which is used in introducing cannulas and similar implements into the body cavities or blood vessels. Trocars are also used as ports in laparoscopic surgeries.
Types of Trocar
- Close Wound Drainage Trocar to be used in Close Wound Suction Unit.
- Thoracic Trocar to be used in Chest Drainage Catheter.
Online Business Query
We will be glad to provide any further information or details that are required. We will be obliged if our company is associated with your eminent organization to cater to the needs of various applications. For customized Trocar, it will be a delight to provide quotations as per the drawing samples with your detailed specification.
Research & Development
Our R&D department is supervised by quality experts who keep a constant eye on the market. Our R&D endeavors comprise:
- Introducing new designs and design control at the time of pre-production state
- Design and development of devices with ease of operation, cutting edge and smoothness that will meet our clients exact specifications
- Close interaction between the production workers and technology teams at all stages.
We use variety of testing and other equipment such as:
- Hardness Tester
- Optical Profile Projector
- Digital Vernier
- Tensile Strength Tester
- Digital Micrometer
- Automatic Vibrator Polishing Unit
- Sand Blasting System (Wet & Dry)
- Boiling Passivation
Chemical Analysis: Mostly done in a metallurgy laboratory on a regular basis.
Standard: Normally, we are following French catheader system or as per clients requirements.
Note: Available from stock with both barbed and threaded connections, wound drainage trocars offered by us are made of premium quality stainless steel that meets ASTM F-899 and ISO 7153-1, all the controlling specifications for stainless steel medical tools and instruments.
Process: We work in accordance to the drawings and other process systems.
Quality Assurance
CONSISTENT QUALITY PRODUCT AT VERY REASONABLE PRICE
We ensure certification of raw material and perform various independent chemical analysis and tensile tests to guarantee consistency of material used. Our products have received appreciable response and acceptance by numerous disposable manufacturers globally. Since the year 2000, we have until now successfully shipped 4 million wound drainage trocars in different sizes to CE approved and ISO certified companies worldwide.
Quality assurance is guaranteed through below mentioned procedures:
- All the inputs are thoroughly checked prior to process line particularly the stainless-steel supplies.
- Rigorous online inspection is done on an hourly basis.
- Intensive testing as well as stage wise inspection
- All the finished goods are thoroughly inspected prior to packing for shipment.
We cater to only medical manufacturers and we maintain full confidentiality during the process of business interaction with our clients.